ANDOVER, Mass., Sept. 9, 2015 /PRNewswire/ -- Reinforcing
its commitment to improving diagnostic confidence through
innovation, Royal Philips (NYSE: PHG AEX: PHIA) today announced
that it has received 510(k) clearance from the U.S. Food and Drug
Administration (FDA) to market its Spectral Diagnostic Suite
(SpDS). Philips SpDS is a set of advanced visualization and
analysis tools designed for the Philips IQon Spectral CT to deliver
enhanced spectral viewing and advanced clinical applications
capabilities.
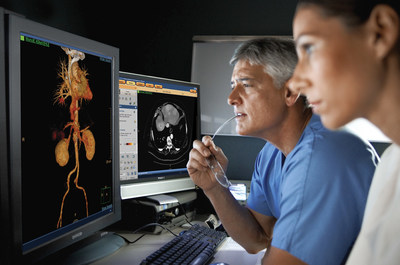
Due to its accessibility, speed and assessment capabilities, CT
imaging is widely used in the diagnosis of many different diseases
and injuries. By providing spectral capabilities within traditional
CT applications, SpDS offers a new level of flexibility and
clinical information for CT users, allowing clinicians to utilize
the spectral information "on-demand," to achieve better clinical
decision support without any added complexity of special modes or
workstations that disrupt user workflow. Additionally, because
there is no need to bring the patient back for additional imaging,
on-demand spectral analysis of a particular region allows the
physician to further analyze incidental findings and at the same
time reduce dose exposure to the patient.
"Philips Spectral suite offers a new level of control for
clinicians, allowing for segmentation on different energy levels,
more detailed comparison between images, and advanced fusion
capabilities, for diagnostic confidence," said Dr. Prabhakar Rajiah, MBBS, M.D., FRCR, Assistant
Professor of Radiology, University Hospitals of Cleveland, UH Case Medical Center, Department
of Radiology, who collaborated with Philips on the development of
the suite.
The Philips SpDS package includes:
- Spectral enhanced Comprehensive Cardiac Analysis (sCCA)
- Spectral enhanced Advanced Vessel Analysis (sAVA)
- Spectral enhanced Tumor Tracking (sTT).
The Philips SpDS also allows for viewing and analysis of
spectral datasets in a variety of settings: a reading room, on
picture archiving and communication systems (PACS), or from remote
locations.
"The game-changing capabilities of the Spectral Diagnostic Suite
represent our ongoing commitment to drive advanced innovation in
medical imaging to improve clinical output and diagnostic
confidence, leading to better care for patients at lower dose
exposure," said Mikael Strindlund, Senior Vice President &
General Manager, CT, for Philips. "Providing clinicians with
additional data to help support the right clinical decisions while
improving workflow and patient experience, is at the core of
everything we do."
For more information on Philips IQon Spectral CT and the Philips
Spectral Diagnostic Suite, please visit:
http://www.spectralctlearningcenter.philips.com/.
About Royal Philips:
Royal Philips (NYSE: PHG, AEX:
PHIA) is a diversified health and well-being company, focused on
improving people's lives through meaningful innovation in the areas
of Healthcare, Consumer Lifestyle and Lighting. Headquartered in
the Netherlands, Philips posted
2014 sales of EUR 21.4 billion and
employs approximately 106,000 employees with sales and services in
more than 100 countries. The company is a leader in cardiac care,
acute care and home healthcare, energy efficient lighting solutions
and new lighting applications, as well as male shaving and grooming
and oral healthcare. News from Philips is located at
www.philips.com/newscenter.
Photo - http://photos.prnewswire.com/prnh/20150908/264515
Logo -
http://photos.prnewswire.com/prnh/20150611/222617LOGO
To view the original version on PR Newswire,
visit:http://www.prnewswire.com/news-releases/philips-receives-fda-510k-clearance-for-spectral-diagnostic-suite-300139404.html
SOURCE Royal Philips