VBL Therapeutics Presents Data on MOSPD2, a Novel Immuno-Oncology Target
April 04 2017 - 7:00AM

VBL Therapeutics (NASDAQ:VBLT), announced today the presentation of
new data on MOSPD2, a novel potential target in oncology. VBL's
study, entitled "MOSPD2, a Newly Characterized Protein, Promotes
Breast Cancer Metastasis" by Mendel et al., will be presented today
at the American Association of Cancer research (AACR) conference in
Washington, DC. The study observed from clinical biopsies that
MOSPD2 is prevalent in invasive human breast cancer tissue and that
levels of MOSPD2 correlate to breast cancer invasiveness. It was
further observed that a knockdown of MOSPD2 in a human breast
cancer cell line using CRISPR technology led to blockade of EGF
signaling and significant reduction of breast cancer cell
migration in vitro and metastasis in a mouse model.
"The current publication indicates involvement of
MOSPD2 in motility and metastasis of cancer cells in a breast
cancer model, with correlative clinical specimens expression
pattern that is associated with breast tumor invasiveness," said
Eyal Breitbart, PhD, VP for Research and Operations at VBL. "We
believe that MOSPD2 may be involved in the regulation of cell
motility in addition to breast cancer, as it is found in other
tumor tissues as well. We recently reported its role in
monocyte migration and are studying its expression and potential
involvement in additional tumor types," added Dr. Breitbart.
The company believes that targeting of MOSPD2 may
have several therapeutic applications, including inhibition of
tumor cell metastases and targeting of MOSPD2-positive tumor cells,
as well as inhibition of monocyte migration in chronic inflammatory
conditions. VBL's "VB-600 series" of pipeline candidates is
being developed towards these applications.
About VBL Vascular Biogenics Ltd.,
operating as VBL Therapeutics, is a clinical stage
biopharmaceutical company focused on the discovery, development and
commercialization of first-in-class treatments for cancer. The
Company’s lead oncology product candidate, ofranergene obadenovec
(VB-111), is a unique biologic agent that uses a dual mechanism to
target solid tumors. It utilizes an angiogenesis-specific sensor
(VBL's PPE-1-3x proprietary promoter) to specifically target the
tumor vasculature, by induction of cell death in angiogenic
endothelial cells in the tumor milieu. Moreover, it is an
immune-stimulant that triggers a local anti-tumor immune response,
which is accompanied by recruitment of CD8 T-cells and apoptosis of
tumor cells. Ofranergene obadenovec is conveniently administered as
an IV infusion once every two months. It has been observed to be
well-tolerated in >200 cancer patients and we have observed its
efficacy signals in an “all comers” Phase 1 trial as well as in
three tumor-specific Phase 2 studies. Ofranergene obadenovec is
currently being studied in a Phase 3 pivotal trial for recurrent
Glioblastoma, conducted under an FDA Special Protocol Assessment
(SPA).
Forward Looking Statements This
press release contains forward-looking statements. All statements
other than statements of historical fact are forward-looking
statements, which are often indicated by terms such as
“anticipate,” “believe,” “could,” “estimate,” “expect,” “goal,”
“intend,” “look forward to”, “may,” “plan,” “potential,” “predict,”
“project,” “should,” “will,” “would” and similar expressions. These
forward-looking statements include, but are not limited to,
statements regarding the clinical development of ofranergene
obadenovec (VB-111) and its therapeutic potential and clinical
results, as well as the potential relevance of MOSPD2 in immune
cells and in motility and metastasis of tumor cells and the
clinical development and therapeutic potential of the VB-600 series
of pipeline candidates. These forward-looking statements are not
promises or guarantees and involve substantial risks and
uncertainties. Among the factors that could cause actual results to
differ materially from those described or projected herein include
uncertainties associated generally with scientific research and
development, clinical trials and related regulatory reviews and
approvals, and the risk that historical clinical trial results may
not be predictive of future trial results. A further list and
description of these risks, uncertainties and other risks can be
found in the Company’s regulatory filings with the U.S.
Securities and Exchange Commission, including in our annual report
on Form 20-F for the year ended December 31, 2016. Existing
and prospective investors are cautioned not to place undue reliance
on these forward-looking statements, which speak only as of the
date hereof. VBL Therapeutics undertakes no obligation to update or
revise the information contained in this press release, whether as
a result of new information, future events or circumstances or
otherwise.
INVESTOR CONTACT:
Michael Rice
LifeSci Advisors, LLC
(646) 597-6979
Vascular Biogenics (NASDAQ:VBLT)
Historical Stock Chart
From Mar 2024 to Apr 2024
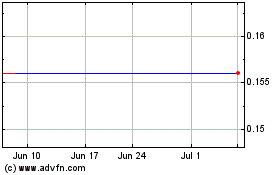
Vascular Biogenics (NASDAQ:VBLT)
Historical Stock Chart
From Apr 2023 to Apr 2024