DUBLIN and ALISO VIEJO, Calif., Sept. 3, 2015 /PRNewswire/ -- Allergan plc
(NYSE: AGN), a leading global pharmaceutical company, and AqueSys,
Inc. a private clinical stage medical device company focused on
developing ocular implants that reduce intraocular pressure (IOP)
associated with glaucoma, today announced that they have entered
into an agreement under which Allergan will acquire AqueSys in an
all-cash transaction. Under the terms of the agreement,
Allergan will acquire AqueSys for a $300
million upfront payment and regulatory approval and
commercialization milestone payments related to AqueSys' lead
development programs, including XEN45.
"The acquisition of AqueSys and its XEN45 program builds on
Allergan's deep and long-standing commitment to innovation in eye
care," said Brent Saunders, CEO and
President of Allergan. "Our eye care team has a strong track
record of introducing novel treatments for patients with glaucoma.
The acquisition of the XEN45 device demonstrates our commitment to
alternative next-generation glaucoma treatments which are appealing
to both patients and physicians and extend beyond conventional
medication eyedrops."
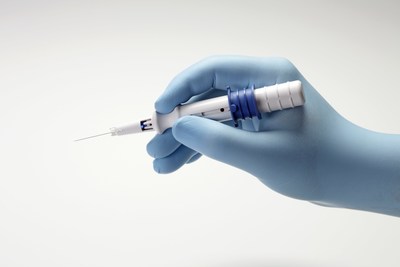
The acquisition of AqueSys adds XEN45, a soft shunt that is
implanted in the subconjunctival space in the eye through a
minimally invasive procedure with a single use, pre-loaded
proprietary injector. The proprietary XEN45 technology facilitates
aqueous fluid flow to lower Intra-Ocular Pressure (IOP) while
protecting against the potential for hypotony (IOP that is too low)
that is associated with current subconjunctival procedures.
The minimally invasive procedure is performed by
ophthalmologists.
XEN45 has received a CE mark in the European Union where
it is indicated for the reduction of intraocular pressure in
patients with primary open angle glaucoma where previous medical
treatments have failed. The CE mark allows treatment in conjunction
with a cataract procedure or as a standalone procedure. XEN45 is
also approved for use in Turkey,
Canada and Switzerland. AqueSys is pursuing reimbursement
in these countries. In the United
States, XEN45 is in late-stage development, with the final
US Investigational Device Exemption (IDE) clinical trial fully
enrolled in the second quarter of 2015. Final approval by the
U.S. Food and Drug Administration is expected by late 2016 or early
2017 via the 510K device pathway.
"The XEN45 program has been shown to provide a robust efficacy
profile with minimal side effects, and if approved in the U.S.,
would provide an exciting new treatment option for patients, and
one that is highly complementary to our ongoing portfolio and
development programs in this critical treatment area," said
David Nicholson, EVP and President,
Global Brands Research and Development at Allergan.
"With its pedigree in the eye care space, its deep commercial
network in eye care professional communities, and its commitment to
being a leading provider of treatments for glaucoma, Allergan was a
natural fit and a compelling partner for us to maximize the
potential for the XEN45 technology platform," said Ron Bache, CEO of AqueSys. "The addition of the
XEN45 technology to Allergan's world-class eye care development and
commercialization organization will propel the potential use of
this best-in-class technology by global ophthalmologists and the
patients that they treat."
"The XEN45 platform is a minimally invasive reinvention of the
sub-conjunctival outflow approach to the treatment of glaucoma, one
used for many years by surgeons," said Robert N.
Weinreb, M.D., Distinguished Professor of Ophthalmology and
Director of the Shiley Eye Institute at the University of California San Diego. "XEN45
provides the advantage of a simpler and less invasive way of
conducting a well-established procedure to treat our glaucoma
patients, and if approved in the U.S., would provide an exciting
new option for ophthalmologists."
Glaucoma is a group of diseases that damage the eye's optic
nerve and can result in vision loss and blindness.[i]
Several large studies have shown that eye pressure is a major risk
factor for optic nerve damage.[i] In open-angle
glaucoma, even though an ocular fluid drainage angle is "open", the
fluid passes too slowly through a meshwork drain.[I]
When the fluid builds up, the pressure inside the eye rises to a
level that may damage the optic
nerve.[I] When the optic nerve is damaged
from increased pressure, open-angle glaucoma-and vision loss may
result.[i] In 2010, 60.5 million people globally
were living with glaucoma.[ii] Given the aging of
the world's population, the number of people living with glaucoma
may increase to nearly 80 million by 2020.[ii]
The transaction is subject to the expiration or termination of
the waiting period under the Hart-Scott-Rodino Antitrust
Improvements Act of 1976. Pending approvals, Allergan
anticipates closing the transaction in the fourth quarter of
2015.
About Allergan
Allergan plc (NYSE: AGN), headquartered in Dublin, Ireland, is a unique, global
pharmaceutical company and a leader in a new industry model –
Growth Pharma. Allergan is focused on developing,
manufacturing and commercializing innovative branded
pharmaceuticals, high-quality generic and over-the-counter
medicines and biologic products for patients around the world.
Allergan markets a portfolio of best-in-class products that
provide valuable treatments for the central nervous system, eye
care, medical aesthetics, gastroenterology, women's health,
urology, cardiovascular and anti-infective therapeutic categories,
and operates the world's third-largest global generics business,
providing patients around the globe with increased access to
affordable, high-quality medicines. Allergan is an industry leader
in research and development, with one of the broadest development
pipelines in the pharmaceutical industry and a leading position in
the submission of generic product applications globally.
With commercial operations in approximately 100 countries,
Allergan is committed to working with physicians, healthcare
providers and patients to deliver innovative and meaningful
treatments that help people around the world live longer, healthier
lives.
For more information, visit Allergan's website at
www.allergan.com.
About AqueSys, Inc.
AqueSys, Inc. is the first ophthalmic device company to focus
exclusively on the treatment of glaucoma by re-envisioning
sub-conjunctival outflow with a minimally invasive stent for the
reduction of intraocular pressure. Backed by more than a decade of
research and recent commercialization in European and Canadian
markets, the safety and efficacy of the company's proprietary
technology supports glaucoma surgeons and patients in the
management of intraocular pressure to arrest the progression of
vision loss associated with glaucoma. Privately held and
headquartered in Southern
California, AqueSys is working to change the treatment of
glaucoma to benefit patients, physicians and payors around the
world through the research, development, manufacturing and
commercialization of its minimally invasive glaucoma stents.
Additional information about the XEN Gel Stent can be found at
www.aquesys.com.
Forward-Looking Statement
Statements contained in this press release that refer to future
events or other non-historical facts are forward-looking statements
that reflect Allergan's current perspective of existing trends and
information as of the date of this release. Except as expressly
required by law, Allergan disclaims any intent or obligation to
update these forward-looking statements. Actual results may differ
materially from Allergan's current expectations depending upon a
number of factors affecting Allergan's business. These factors
include, among others, the difficulty of predicting the timing or
outcome of FDA approvals or actions, if any; the impact of
competitive products and pricing; market acceptance of and
continued demand for Allergan's products; risks associated with
acquisitions, mergers and joint ventures; difficulties or delays in
manufacturing; and other risks and uncertainties detailed in
Allergan's periodic public filings with the Securities and Exchange
Commission, including but not limited to Allergan's Quarterly
Report on Form 10-Q for the quarter ended June 30, 2015. Except as expressly required by
law, Allergan disclaims any intent or obligation to update these
forward-looking statements.
[i] National Eye Institute – Glaucoma Facts:
https://nei.nih.gov/health/glaucoma/glaucoma_facts
[ii] Bright Focus Foundation – Glaucoma Facts &
Figures:
http://www.brightfocus.org/glaucoma/article/glaucoma-facts-figures
|
CONTACTS:
|
Allergan:
|
|
Investors:
|
|
Lisa
DeFrancesco
|
|
(862)
261-7152
|
|
|
|
Media:
|
|
Mark
Marmur
|
|
(862)
261-7558
|
Photo - http://photos.prnewswire.com/prnh/20150902/263398
Logo - http://photos.prnewswire.com/prnh/20150612/222796LOGO
To view the original version on PR Newswire,
visit:http://www.prnewswire.com/news-releases/allergan-to-acquire-glaucoma-treatment-company-aquesys-to-add-minimally-invasive-implantable-shunt-to-eye-care-offering-300137935.html
SOURCE Allergan plc