BioMarin to Host First Quarter 2015 Financial Results Conference Call and Webcast on Thursday, April 30 at 4:30pm ET
April 14 2015 - 8:00AM

BioMarin Pharmaceutical Inc. (Nasdaq:BMRN) today announced that
Jean-Jacques Bienaimé, Chief Executive Officer of BioMarin, will
host a conference call and webcast on Thursday, April 30, at 4:30
p.m. ET to discuss first quarter 2015 financial results and provide
a general business update.
Dial-in Number U.S. / Canada Dial-in Number:
(877) 303-6313 International Dial-in Number: (631) 813-4734
Conference ID: 10256447
Replay Dial-in Number: (855) 859-2056 Replay International
Dial-in Number: (404) 537-3406 Conference ID: 10256447
Interested parties may access a live audio webcast of the
conference call via the investor section of the BioMarin
website, www.BMRN.com. A replay of the call will be archived
on the site for one week following the call.
About BioMarin
BioMarin develops and commercializes innovative
biopharmaceuticals for serious diseases and medical conditions. The
company's product portfolio comprises five approved products and
multiple clinical and pre-clinical product candidates. Approved
products include Vimizim® (elosulfase alfa) for MPS IVA, a product
wholly developed and commercialized by BioMarin; Naglazyme®
(galsulfase) for MPS VI, a product wholly developed and
commercialized by BioMarin; Aldurazyme® (laronidase) for MPS I, a
product which BioMarin developed through a 50/50 joint venture with
Genzyme Corporation; Kuvan® (sapropterin dihydrochloride) Powder
for Oral Solution and Tablets, for phenylketonuria (PKU), developed
in partnership with Merck Serono, a division of Merck KGaA of
Darmstadt, Germany; and Firdapse® (amifampridine), which has been
approved by the European Commission for the treatment of Lambert
Eaton Myasthenic Syndrome (LEMS). Product candidates include
drisapersen, an exon skipping oligonucleotide, which is currently
undergoing regulatory submission for the treatment of Duchenne
muscular dystrophy (exon 51); pegvaliase (PEGylated recombinant
phenylalanine ammonia lyase, formerly referred to as BMN 165 or PEG
PAL), which is currently in Phase 3 clinical development for the
treatment of PKU; talazoparib (formerly referred to as BMN 673), a
poly ADP-ribose polymerase (PARP) inhibitor, which is currently in
Phase 3 clinical development for the treatment of germline BRCA
breast cancer; reveglucosidase alfa (formerly referred to as BMN
701), a novel fusion protein of insulin-like growth factor 2 and
acid alpha glucosidase (IGF2-GAA), which is currently in Phase 3
clinical development for the treatment of Pompe disease; BMN 111, a
modified C-natriuretic peptide, which is currently in Phase 2
clinical development for the treatment of achondroplasia; BMN 044
and BMN 045, exon skipping oligonucleotides, which are currently in
Phase 2 clinical development for the treatment of Duchenne muscular
dystrophy (exons 44 and 45); BMN 053, an exon skipping
oligonucleotide, which is currently in Phase 1/2 clinical
development for the treatment of Duchenne muscular dystrophy (exon
53); cerliponase alfa (formerly referred to as BMN 190), a
recombinant human tripeptidyl peptidase-1 (rhTPP1), which is
currently in Phase 1/2 clinical development for the treatment of
CLN2 disorder, a form of Batten disease; BMN 270, an AAV-factor
VIII vector, for the treatment of hemophilia A; and BMN 250, a
novel fusion of alpha-N-acetyglucosaminidase (NAGLU) with a peptide
derived from insulin-like growth factor 2 (IGF2), for the treatment
of MPS IIIB.
For additional information, please visit www.BMRN.com.
Information on BioMarin's website is not incorporated by reference
into this press release.
CONTACT: Investors
Traci McCarty
BioMarin Pharmaceutical Inc.
(415) 455-7558
Media
Debra Charlesworth
BioMarin Pharmaceutical Inc.
(415) 455-7451
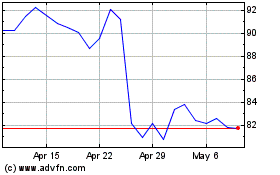
BioMarin Pharmaceutical (NASDAQ:BMRN)
Historical Stock Chart
From Mar 2024 to Apr 2024
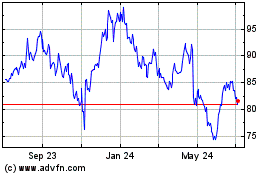
BioMarin Pharmaceutical (NASDAQ:BMRN)
Historical Stock Chart
From Apr 2023 to Apr 2024